By Ben Fuchs | Pharmacist Ben
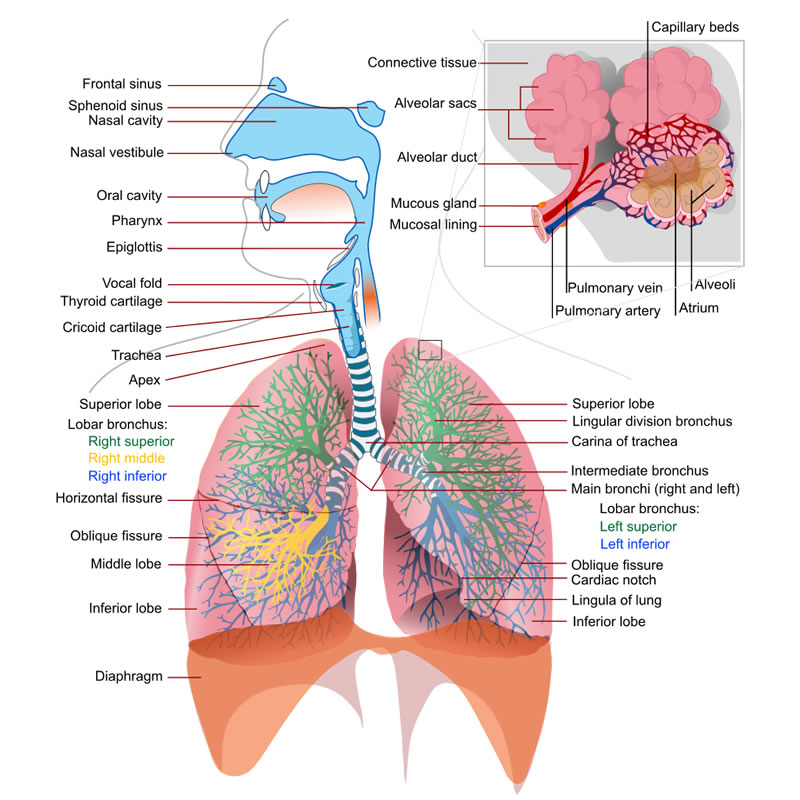
I was watching a show on the history channel over the weekend. It was called The Story of US and they were talking about Andrew Carnegie and the development of the Steel industry. Our whole external outside world is structured by steel in skyscrapers and ships and cars and machines and appliances. It’s really amazing stuff, and all it is iron that been reacted with oxygen. Oxygen makes things happen. It makes things hotter. Our air is about 14 or 15 percent oxygen. If it were any higher, our environment would become very unstable and there would be lots of explosion. Oxygen makes things happen! For better or worse. An apple exposed to oxygen turn brown and iron nail will turn orange.

The same History Channel show talked about the Statue of Liberty. If you’ve ever seen that Statue of Liberty or a picture of it can see that the Statue of Liberty has this interesting green color. Well apparently that wasn’t the color of Statue of Liberty when she was first put up. That green color is the reaction of the copper to…oxygen! Oxygen makes things happen. That’s how peroxide works. Per-oxide refers to 2 pieces of oxygen. In fact, hydrogen per-oxide is nothing more than plain old water with a piece of oxygen stuck on it. Water is H2O and Hydrogen Peroxide is H2O2. That extra oxygen makes peroxide much more active than water. Oxygen makes things happen. And as soon as the oxygen is releases H202 turns into plain old H2O again and that’s why hydrogen peroxide is so safe. But only at low concentration, because oxygen makes things happen. It’s active. So food grade Hydrogen Peroxide is only around 12- 35 percent AND YOU PROBABLY want to dilute that. The antiseptic that so effectively kills germs is 3% hydrogen peroxide.